Curriculum Vitae
| Carrier | Latvian Institute of Organic Synthesis, Riga, Latvia 2021 Principal Researcher 2020 Researcher 2007 Research Assistant 2006 Laboratory Assistant University of Latvia, Faculty of Chemistry, Riga, Latvia 2003 – 2005 Senior Laboratory Assistant |
| Education | 2023 (03) University of Florence, Italy (Prof. C. T. Supuran) 2022 (09-11) HMX Pro Harvard medical School, Certificates of Achievement in Pharmacology; Drug Discovery and Development; Drug Delivery 2022 (05-07) Montpellier University, France (Prof. J.-Y. Winum group) 2019 Dr. Chem., Faculty of Chemistry, University of Latvia 2018 (05-08) PELICO International Project, Enamine Ltd., Ukraine 2016 (04-08) InnovaBalt Exchange Program, University of Oxford, UK (Prof. J. Robertson) 2015 (02-05) InnovaBalt Exchange Program, Universita di Parma, Italy (Prof. G. Costantino) 2007 M.Sc. Chem., Faculty of Chemistry, University of Latvia 2006 Master 2, Universite de Nantes, Faculte Pharmaceutique, France 2004 B.Sc. Chem., Faculty of Chemistry, University of Latvia |
| Awards | 2014 Poster Award, Balticum Organicum Syntheticum, Lithuania 2013 ESMEC Fellowship, 33th Summer School of Medicinal Chemistry, Urbino, Italy |
List of Publications
10. Design, Synthesis, and Bioactivity Evaluations of 3-Methylenechroman-2-one Derivatives as Thioredoxin Reductase (TrxR) Inhibitors
Nikitjuka, A.; Ozola, M.; Krims-Davis, K.; Žalubovskis, R. ChemMedChem, 2023, e202300504. DOI:10.1002/cmdc.202300504

9. Boron-containing carbonic anhydrases inhibitors
Giovannuzzi, S.; Nikitjuka, A.; Pereira Resende, B. R.; Smietana, M.; Nocentini, A.; Supuran, C. T.; Winum, J.-Y. Bioorg. Chem., 2024, 143, 106976. DOI:10.1016/j.bioorg.2023.106976

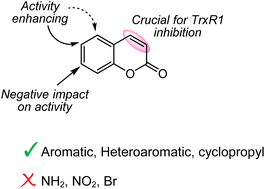
8. Exploration of 3,4-unsubstituted coumarins as thioredoxin reductase 1 inhibitors for cancer therapy
Nikitjuka, A.; Ozola, M.; Jackevica, L.; Bobrovs, R.; Žalubovskis, R. Org. Biomol. Chem., 2023, 21, 9630-9639. DOI:10.1039/D3OB01522J
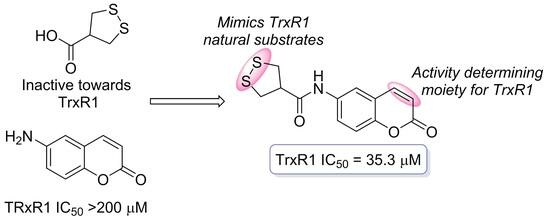
7. May 1,2-Dithiolane-4-carboxylic Acid and Its Derivatives Serve as a Specific Thioredoxin Reductase 1 Inhibitor?
Nikitjuka, A.; Krims-Davis, K.; Kanepe-Lapsa, I.; Ozola, M.; Žalubovskis, R. Molecules, 2023, 28, 6647. DOI:10.3390/molecules28186647
6. Asparagusic Acid – A Unique Approach toward Effective Cellular Uptake of Therapeutics: Application, Biological Targets, and Chemical Properties
Nikitjuka, A.; Žalubovskis, R. ChemMedChem, 2023, e202300143. DOI:10.1002/cmdc.202300143

5. Refining the Structure−Activity Relationships of 2-Phenylcyclopropane Carboxylic Acids as Inhibitors of O-Acetylserine Sulfhydrylase Isoforms
Magalhães, J.; Franko, N.; Annunziato, G.; Pieroni, M.; Benoni, R.; Nikitjuka, A.; Mozzarelli, A.; Bettati, S.; Karawajczyk, A.; Jirgensons, A.; Campanini, B.; Costantino, G. J. Enzyme Inhib. Med. Chem., 2019, 34, 31-43. DOI:10.1080/14756366.2018.1518959

4. Synthesis and Biological Evaluation of Aziridin-1-yl Oxime-based Vorinostat Analogs as Anticancer Agents
Nikitjuka, A.; Shestakova, I.; Romanchikova, N.; Jirgensons A. Chem. Heterocycl. Compd., 2015, 51(7), 647-657. DOI:10.1007/s10593-015-1752-z

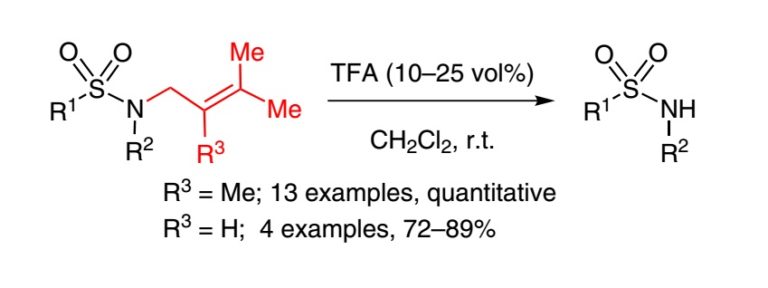
3. Methylprenyl and Prenyl Protection for Sulfonamides
Nikitjuka, A.; Nekrasova, A.; Jirgensons A. Synlett, 2015, 26, 183-186. DOI:10.1055/s-0034-1379428
2. Synthesis, Chemical and Biological Properties of Aziridine-1-carbaldehyde Oximes (Minireview)
Nikitjuka, A.; Jirgensons, A. Chem. Heterocycl. Compd., 2014, 49, 1544-1559. Dedicated to Professor Janis Stradins on the occasion of his 80 anniversary. DOI:10.1007/s10593-014-1407-5
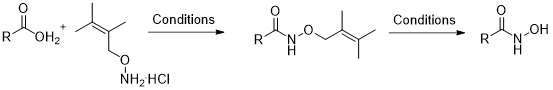
1. Synthesis of Hydroxamic Acids by Using the Acid Labile O-2-Methylprenyl Protecting Group
Nikitjuka, A; Jirgensons, A. Synlett, 2012, 23, 2972-2974. DOI:10.1055/s-0032-1317687